|
Imaging technology today uses a p-polarized HeNe laser beam as a light source and the reflected light is directed at a CCD camera. This demonstrates the basic principle that the energy is transferred to the ligands and biomolecules which in turn, change the amount of reflectivity of light and reflect back a gradient of absorbed spectra that helps to understand the composition of the material. A very simple form of this is shown in Figure 4. This transfer of energy through the material, allows for SPR imaging. 
The SPR allows certain frequencies of light through, allowing for an image of how the biomolecules are attached to the ligands. Similar to the other electron displacement effect, mesomeric effect is also classified into positive mesomeric effect ( M or R) and negative mesomeric effect (-M or -R) based on the nature of the functional group present adjacent to the multiple bonds.\): : A simple schematic representation of how light enters the prism, goes through the thin metal film where SPR is occuring on the side of the film touching the ligands and biomolecules. The resonance hybrid is represented as below. The resonating structures are called canonical forms and the actual structure lies between these three resonating structures, and is called a resonance hybrid. Equivalent Lewis structures are called resonance forms. Actually, the n electrons are delocalized as shown below. Last updated A resonance form is another way of drawing a Lewis dot structure for a given compound. Each resonating structure contributes to real. Resonance is a mental exercise and method within the Valence Bond Theory of bonding that describes the delocalization of electrons within molecules. The resonance hybrid is imaginary structure which is obtained by mental combination of resonating structures. This property cannot be explained by a simple structure in which two n bonds localized between C 1 – C 2 and C 3 – C 4. Some molecules have two or more chemically equivalent Lewis electron structures, called resonance structures. A resonance structure is one of multiple Lewis diagrams for the same molecule. Two structures of a given molecule, in which the components are the same but the electron placements differ are said to be resonance structures of one another. What is a resonance structure in chemistry. In 1,3 butadiene, it is expected that the bond between C 1 – C 2 and C 3 – C 4 should be shorter than that of C 2 – C 3, but the observed bond lengths are of the same. Because of the presence of double bonds or pi bonds in a chemical compound, it is difficult to localize the position of all electrons. The resonance effect in organic chemistry is the variation in electron behavior when elements other than hydrogens and carbons take part actively in the. 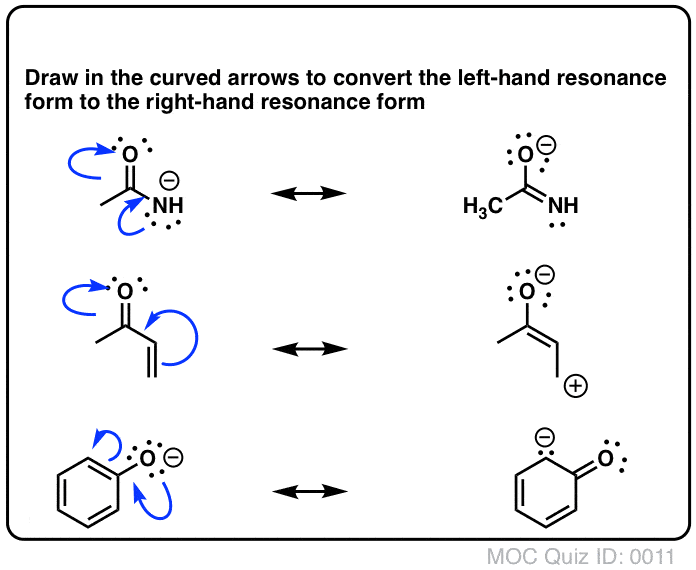
This phenomenon is also called mesomerism or mesomeric effect.įor example, the structure of aromatic compounds such as benzene and conjugated systems like 1,3 – butadiene cannot be represented by a single structure, and their, observed properties can be explained on the basis of a resonance hybrid. Such structures are called resonance structures (canonical structures) and this phenomenon is called resonance. Certain organic compounds can be represented by more than one structure and they differ only in the position of bonding and lone pair of electrons. Resonance is a way of describing delocalized electrons within certain molecules or polyatomic ions where the bonding cannot be expressed by a single Lewis formula. The resonance is a chemical phenomenon that is observed in certain organic compounds possessing double bonds at a suitable position. Magnetic Resonance in Chemistry is devoted to the rapid publication of papers which are concerned with the development of magnetic resonance techniques. Resonance arises when more than one valid Lewis structure can be drawn for a molecule or ion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
